Nachfolgend wollen wir einen kurzen Überblick über das Wesen der Hydrophobierung in der Bauchemie bzw. für bauchemische Anwendungen geben.
Hydrophobierung - was ist das?
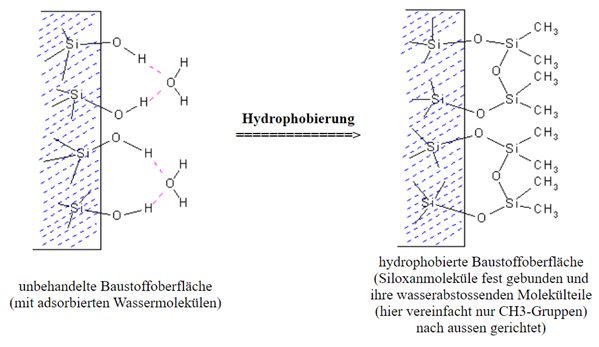
Die Hydrophobierung von Baustoffen bedeutet, diesen wasserabweisend zu machen bzw. wasserabweisend auszustatten. Der hydrophobierende Wirkstoff besetzt hierbei die Poren- und Kapillaroberflächen und macht sie dadurch wasserabweisend. Kapillaren und Poren werden dabei jedoch nicht komplett verschlossen, sodass die Atmungsaktivität (Diffusionsfähigkeit) des jeweiligen Werkstoffes erhalten und bestehen bleibt.
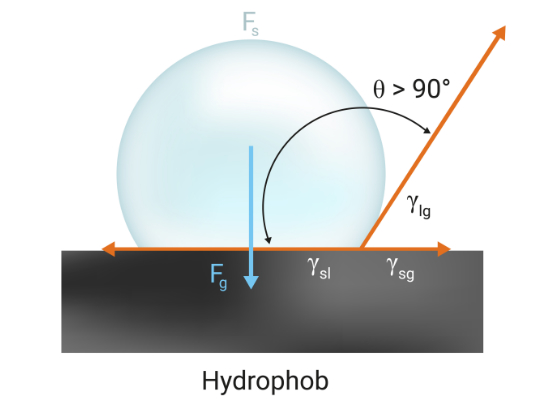
Abb: Ist der Winkel > 90°, so ist die Oberfläche wasserabweisend, also hydrophob.
Hydrophobierungsmittel auf Basis organischer Siliciumverbindungen benötigen zur Reaktion zum endgültigen Wirkstoff (Silikonharz) immer auch Feuchtigkeit, diese ist in jedem Baustoff bzw. Substrat jedoch in ausreichender Menge zur Verfügung.
Um die Wirksamkeit optimal entfalten zu können, muss jedenfalls gewährleistet sein, dass die Silikonharzmolekühle an der Werkstoffoberfläche fest gebunden werden und auch eine entsprechende Eindringtiefe in das Substrat gegeben ist.
Quelle: Andrea Glatthor -
Baustoffchemie.de
Übersicht über die verschiedenen Wirkstoffe (org. Si-Verbindungen)
1. Alkoxysilane (vereinfacht oft "Silane" genannt)
Unter Silanen werden im Bautenschutz die Alkylalkoxysilane verstanden, während "anhydrolisierte Silane" wiederum eher zu den Alkylalkoxysiloxanen gehören. Varianten:
- gelöst in wasserfreien Alkoholen (z.B. Isopropanol)
- gelöst in aliphatischen Lösemitteln
- emulgiert in Wasser (z.B. das Triethoxyoctylsilan)
2. Alkoxysiloxane (oft einfach nur "Siloxane" genannt)
Es gibt oligomere und polymere Siloxane, sprich: Produkte mit kleinen und großen Molekülen. Varianten:
- Gelöst in aliphatischen Lösemitteln.
- Emulgiert in Wasser.
- Als Microemulsionskonzentrat (Silane und oligomere Alkoxysiloxane im Gemisch). Bei Silicon-Mikroemulsionskonzentraten (SMK) handelt es sich um ein 100%iges Silikonprodukt, das mit Wasser verdünnbar ist und dabei spontan eine Microemulsion bildet.
3. Silikonharze (Alkylpolysiloxane)
4. Alkalisiliconate
Das sind hochalkalische Lösungen von Kaliumsiliconat. Auf Baustoffe aufgetragen reagieren sie mit dem Kohlendioxid der Luft, wodurch Silanol als Zwischenstufe entsteht und dies weiter zum Silikonharz reagiert. Dabei entsteht Kaliumcarbonat (Pottasche, K2CO3) als Nebenprodukt, das sich dann als weißer Belag auf der Oberfläche störend bemerkbar machen kann und speziell bei Fassaden stören würde.
Angewendet werden die Produkte vorwiegend zur werkseitigen Imprägnierung von Ziegeln, Gasbeton und ähnlichen Baustoffen, sind aber auch vielfach in Injektagemitteln für nachträgliche Horizontalsperren enthalten (dort meist in Kombination mit Wasserglas). Zu empfehlen sind diese Produkte nur für die nachträgliche Imprägnierung von Gipsbaustoffen (wenn z.B. irrtümlich "weiße" Gipskartonplatten eingebaut wurden, anstatt der vorimprägnierten "grünen" oder nur Teilbereiche hydrophobiert werden sollen).
Alle vier Wirkstoffe ergeben letztendlich ein Silikonharz als Reaktionsprodukt auf der Baustoffoberfläche:
Reaktion mit Feuchtigkeit
Silan -----------------------------------> Silikonharz
Molekülvergrößerung
Reaktion mit Feuchtigkeit
Siloxan -----------------------------------> Silikonharz
Molekülvergrößerung
Verdunsten des Lösungsmittels
Silikonharz -----------------------------------> Silikonharz
Reaktion mit CO2 der Luft
Silikonat -----------------------------------> Silikonharz
Molekülvergrößerung
Unabhängig davon, welchen Typ Hydrophobierungsmittel man verwendet, entsteht bei allen als Endprodukt ein Silikonharz - die Molekülgröße dieses Silikonharzes kann jedoch sehr unterschiedlich sein, je nachdem, welche der genannten Ausgangsstoffe man einsetzt. Das ist hier nur von untergeordneter Bedeutung, soll aber nicht unerwähnt bleiben. Die gesamte Darstellung hier wurde stark vereinfacht.
5. Hilfsstoffe
Hilfsstoffe umfassen sowohl Lösemittel wie z. B. Isoparaffine, div. Benzine und Spezialbenzine wie auch Alkohole. Weiters diverse Additive wie Netzmittel, Konservierungsmittel, Emulgatoren und Katalysatoren.
Übersicht über die wichtigsten Zubereitungsarten
1. Imprägnier-Emulsionen, wässrig
Lösemittelfreie, wasserbasierte Emulsionen aus Kombinationen von Silanen und/oder Siloxanen. Es gibt auch wasser- und lösemittelfreie Konzentrate auf dem Markt, die vor der Verarbeitung erst mit Wasser verdünnt und dabei emulgiert werden (sogen. Mikroemulsionen).
2. Imprägnier-Paste
Hochviskose, also thixotrope Emulsionen von Siloxanen und Silanen in Wasser. Durch die hohe Viskosität kann die Creme mittels Rolle, Bürste oder Airless-Spritze aufgebracht werden, ohne dass sie abläuft.
3. Imprägnier-Lösungen, lösungsmittelhaltig
Hochalkylierte Alkoxysiloxane, die sich weder in Alkoholen noch in Wasser lösen oder emulgieren lassen. Sie lösen sich nur in Benzin oder ähnlichen organischen Lösungsmitteln und sind bereits ca. 1970 Stand der Technik gewesen.
4. Imprägnier-Lösungen, wässrig
Wenn das Verkaufsprodukt eine wässrige Lösung ist, handelt es sich in den meisten Fällen um Kalium-Methylsilikonate oder Kalium-Propylsilikonate. Diese haben einen recht eingeschränkten Anwendungsbereich. Sie sind die ersten siliciumorganischen Verbindungen auf dem Markt gewesen (ca. 1955).
Grundsätzlich gibt es die Möglichkeit die jeweiligen Hydrophobierungsmittel entweder:
- In wässriger Lösung bzw. in Wasseremulgiert oder
- In Lösemitteln formuliert (wasserverdünnbar oder auch lösemittelverrdünnbar) einzusetzen. Die Unterschiede liegen in der Wirksamkeit der verschiedenen Produkte und Zubereitungen sowie in der Eindringtiefe, Reaktion, Art des Untergrundes, ezc begründet und müssen vor der Hydrophobierung genau getestet werden.
Die richtige Anwendung diverser Hydrophobierungsmittel
Ziel bei der Anwendung diverser Hydrophobierungsmittel muss es sein eine optimale Wirksamkeit mit so wenig wie möglich ungewollten Nebeneffekten zu erzielen. Grundsätzlich ist die Wirksamkeit eines Hydrophobierungsmittels umso besser, je besser die Eindringtiefe ist. liegt die optimale Eindingtiefe bei normalen Fassaden jedoch nur bei 2 bis 4 mm, während bei Flächen, die einer Frost-Tausalzbeanspruchung ausgesetzt sind (waagerechte Flächen aus Beton , Estrich oder Pflastersteinen), die optimale Eindringtiefe deutlich höher ist (6-8 mm).
Die Eindringtiefe ist von verschiedenen Faktoren abhängig:
- der Saugfähigkeit des zu imprägnierenden Baustoffes und dessen Feuchtegehalt zum Zeitpunkt der Imprägnierung (ebenso dessen chemischer Aufbau, da die chemisch-physikalischen Wechselwirkungen zwischen Baustoff und Wirkstofflösung ganz entscheidende Bedeutung haben)
- der Art des Wirkstoffes (u.a. der Größe der Moleküle, aber auch die chemische Struktur, wegen der eben erwähnten Wechselwirkungen)
- der Konzentration des Wirkstoffes (ein üblicher Wirkstoffgehalt ist 5%; zu viel Wirkstoff schadet eher)
- der Art des Lösungsmittels (je nach Wirkstoff und Untergrundgegebenheiten wechselt das optimale Lösungsmittel)
- der Anwendungstechnik (der Verarbeitungsweise, der Auftragsmenge, der Kontaktzeit, der Anzahl der einzelnen Arbeitsgänge und der Zeitabstände zwischen ihnen
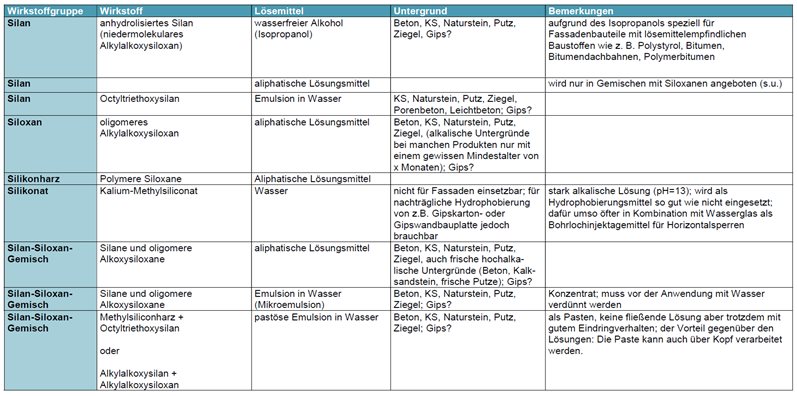
Produktgruppen/Stoffklassen
Fazit: Schutz und Verlängerung der Lebensdauer
Die Hydrophobierung von Baustoffen und Baumaterialien schützt nicht nur das Material an sich, sondern auch die Wärmedämmung wird verbessert, wodurch es zur Einsparung von Energiekosten kommt.
Das
Produktportfolio der Donauchem GmbH umfasst eine Reihe von Hydrophobierungsmittel.
Portfolio Additive
Portfolio Paint & Construction
Für weitere Informationen oder Angebote
kontaktieren Sie uns bitte.
 www.donauchem.at
Weiterführende Links/Quellen:
Baustoffchemie.de
Dichtstoffhandel.de
www.donauchem.at
Weiterführende Links/Quellen:
Baustoffchemie.de
Dichtstoffhandel.de