Hinter den Kulissen der Donauchem: Wie Qualität und Sicherheit in der Distribution und Lohnproduktion Hand in Hand gehen
Die chemische Industrie unterliegt zahlreichen gesetzlichen Vorschriften und branchenspezifischen Normen. Eine effektive Qualitätskontrolle und Qualitätssicherung gewährleistet die Einhaltung dieser Vorgaben und ist entscheidend, um eine hohe Produktqualität und die Zufriedenheit unserer Kunden sicherzustellen. Oliver Groß, Leiter der Qualitätssicherung der Donauchem, gibt Ihnen Einblicke in die zentralen Aufgaben und Abläufe der Qualitätskontrolle und Qualitätssicherung bei der Donauchem.
Qualitätsmanagement, Qualitätskontrolle, Qualitätssicherung – was ist der Unterschied?
Ein passender Vergleich zur Abgrenzung dieser Begrifflichkeiten sind die Komponenten eines Fahrrads:
Während das Qualitätsmanagement den Lenker darstellt und die strategische Ausrichtung vorgibt, sind die produktbezogene Qualitätskontrolle und die prozessorientierte Qualitätssicherung die beiden Räder, die das System in Bewegung halten:
- Die Qualitätskontrolle bezieht sich speziell auf die Überprüfung der Produktqualität, indem Proben entnommen und verschiedenen Tests unterzogen werden.
Es existiert ein detaillierter Probenplan, der spezifische Analyseparameter festlegt und vordefinierte Geräte vorschreibt, auf denen die Tests durchgeführt werden. Die Schritte dieses Prozesses sind genau strukturiert und definiert, um sicherzustellen, dass alle Aspekte der Qualitätsprüfung effizient und präzise ausgeführt werden.
- Die Qualitätssicherung legt hingegen die Prozesse, Verfahren und Methoden fest, die benötigt werden, um die Qualität entlang der gesamten Wertschöpfungskette der Donauchem zu sichern.
- Zusätzlich überwacht und überprüft das Qualitätsmanagement alle Bereiche die sich direkt oder indirekt auf die Kundenzufriedenheit auswirken, implementiert Verfahren zur Risikominderung, Überwacht die Schulung von Mitarbeitern sowie die Durchführung von internen Audits, um die Einhaltung der Qualitätsstandards sicherzustellen.
Eine klare Trennung dieser Aufgaben ist bei der Donauchem nicht immer möglich, da die Übergänge fließend sind und die Qualitätskontrolle auch Aspekte der Qualitätssicherung die Qualitätssicherung Teilbereiche vom Qualitätsmanagement übernimmt. Alle drei Bereiche arbeiten eng zusammen, um eine konstant hohe Produktqualität zu gewährleisten. Der Fokus nachfolgender Ausführungen liegt vor allem auf der Qualitätskontrolle.
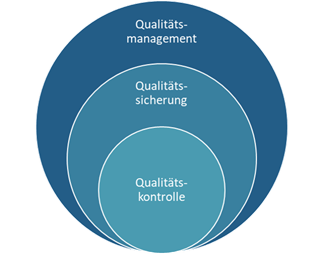
Abbildung 1: QM, QS und QK arbeiten in Synergie, um die Qualität in der chemischen Distribution und Lohnproduktion der Donauchem zu gewährleisten.
Gesetzliche Vorschriften und Normen: Fundament der Qualitätskontrolle
Die Aufgaben der Qualitätskontrolle sind äußerst vielfältig und anspruchsvoll. Ein zentraler Bestandteil besteht in der sorgfältigen Überprüfung der Produktqualität anhand der spezifischen Anforderungen, die nicht nur in den geltenden Gesetzen und Normen festgelegt sind, sondern auch in den individuellen Vereinbarungen mit unseren Kunden.
1. Gesetzliche Bestimmungen
In bestimmten Produktbereichen, wie beispielsweise bei Lebensmitteln und Lebensmittelzusatzstoffen, gelten EU-Regularien, die spezifische Parameter und Anforderungen festlegen, die als Mindest-Qualitätsstandards eingehalten werden müssen.
Ähnlich verhält es sich bei Arzneimitteln und deren Vorstufen, die im Europäischen Arzneibuch angeführt werden. In Österreich wird neben dem österreichischen Arzneibuch (ÖAB) das EU-Arzneibuch (Ph.Eur.) als anerkannte Referenz für die Qualität und Standards von Arzneimitteln verwendet.
Bei technischen Chemikalien lässt der Gesetzgeber den Spielraum für die Definition von Qualitätsstandards hingegen relativ weit offen. Spezifikationen können je nach Bedarf und Anwendung festgelegt werden.
Das bedeutet, dass die Qualität der Produkte von Pharma-Qualität bis hin zu technischer Qualität einer großen Spanne unterliegt und individuell mit unseren Kunden abgestimmt wird.
2. Relevante Normen
Um sicherzustellen, dass hohe Qualitätsstandards eingehalten werden, hat die Donauchem bereits im Jahr 1995 ein umfassendes Qualitätsmanagementsystem implementiert. Dieses System umfasst alle Standorte des Unternehmens und ist gemäß der aktuellen Version der ISO 9001 zertifiziert. Zusätzlich zu dieser Zertifizierung haben wir weitere Zertifizierungen in verschiedenen Bereichen erworben:
- ISO 22000: Diese Norm befasst sich speziell mit Lebensmittelsicherheit und stellt sicher, dass die entsprechenden Anforderungen und Kontrollmechanismen implementiert sind, um die Sicherheit der hergestellten Lebensmittel zu gewährleisten.
- Responsible Care®: Die Donauchem beteiligt sich an der Responsible Care-Initiative, einer globalen Initiative der chemischen Industrie, die sich auf Umweltschutz, Gesundheit und Sicherheit konzentriert.
- RSPO: Die Donauchem bezieht Palmölprodukte aus nachhaltig bewirtschafteten Palmplantagen, die gemäß den Standards des Roundtable on Sustainable Palm Oil (RSPO) zertifiziert sind.
Standort
|
Zertifiziert nach
EN ISO 9001
i.d.g.F. |
Zertifiziert nach
ISO 22000
(Lebensmittel) |
Zertifiziert nach
Responsible Care
|
Zertifiziert nach
den Kriterien
RSPO |
| Donauchem |
seit 1995 |
|
|
|
Standort Brückl
(Errichtung 2022) |
seit 2022
|
seit 01/2023
|
in Planung
|
|
Standort Pischelsdorf
(Errichtung 2008) |
seit 2008
|
seit 2019
(davor HACCP) |
seit 2015
|
seit 2019
|
Abbildung 2: Insgesamt 15 interne Auditoren und Auditorinnen mit unterschiedlichen Spezialisierungen in den Bereichen Qualitätssicherung, Umwelt, Hygiene und Sicherheit sorgen dafür, dass die Standards intern eingehalten werden.
3. Individuelle Qualitätsvereinbarungen
Die Qualitätskontrolle überwacht nicht nur die Einhaltung gesetzlicher Vorschriften und Normen, sondern auch individuelle Qualitätsvereinbarungen mit den Kunden. Diese Vereinbarungen können spezifische Anforderungen enthalten, die über die reinen Spezifikationen hinausgehen.
Ein Beispiel hierfür ist die Kennzeichnung von Produkten. Wenn der Kunde beispielsweise eine Lieferung mit einer roten Markierung wünscht, liegt es in der Verantwortung der Qualitätskontrolle sicherzustellen, dass diese Kennzeichnung korrekt durchgeführt wird.
Darüber hinaus können Kunden besondere Lieferanforderungen haben, wie die Bereitstellung eines externen Zertifikats. In solchen Fällen wird von Mitarbeitern der Qualitätskontrolle ein Muster gezogen, das an eine externe Stelle gesendet und analysiert wird, um die erforderlichen Dokumente für die Lieferung bereitzustellen.
Die Qualitätskontrolle spielt somit eine entscheidende Rolle bei der Erfüllung individueller Kundenanforderungen und stellt sicher, dass alle Aspekte der Qualität gemäß den getroffenen Vereinbarungen erfüllt werden.
Die drei Phasen der Qualitätskontrolle
Der Qualitätskontrollprozess ist in drei eigenständige Prozesse unterteilt: Qualitätskontrolle beim Wareneingang, während der Produktion und beim Warenausgang. Jedes einzelne Produkt durchläuft einen akribischen Analyseprozess, bei dem wir sicherstellen, dass es den vorgegebenen Qualitätsrichtlinien und vereinbarten Qualitätsanforderungen entspricht.
1. Wareneingangskontrolle
Bei der Eingangskontrolle wird die Ware bei ihrer Ankunft gründlich kontrolliert und die zugehörigen Zertifikate werden geprüft. Im Vordergrund steht die zügige Beurteilung oder nach Notwendigkeit die Bemusterung und Analyse grundlegender Parameter um die Identität und die Qualität der angelieferten Ware zu überprüfen. Bei Tankwägen sind die Proben bevorzugt zu behandeln, damit der Tank möglichst rasch entladen werden kann.
Im Zuge der Qualitätskontrolle überprüfen wir jede Kammer des Tankwagens und betrachten dabei repräsentative physikalische Parameter wie Gehalt, Dichte, Brechungsindex oder zum Beispiel auch den Wassergehalt bei Lösemitteln.
Anhand dieser Parameter können wir rasch feststellen, ob die Ware unseren Spezifikationen entspricht und für die Verarbeitung, Lagerung oder den Weitertransport freigegeben werden kann.
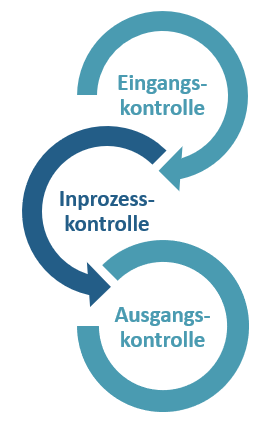
2. Inprozesskontrolle
Inprozesskontrollen sind ein wesentlicher Bestandteil des Produktionsprozesses und dienen dazu, sicherzustellen, dass das in Herstellung befindliche Produkt den festgelegten Spezifikationen entspricht.
Die Probeentnahmen werden während der laufenden Produktion durchgeführt und sind insbesondere bei komplexen Mischungen relevant, bei denen bestimmte Parameter eingestellt werden müssen.
# Überprüfung und Anpassung von Parametern
Ein wichtiger Output der Inprozesskontrolle besteht darin, Abweichungen von Grenzwerten in den festgelegten Parametern zu identifizieren und zu korrigieren. In solchen Fällen ist es entscheidend, die Ursachen zu ermitteln und zu untersuchen, ob möglicherweise falsche Spezifikationswerte vorliegen.
Nehmen wir zum Beispiel eine Mischung, bei der ein bestimmter pH-Wert erreicht werden soll. Wenn dieser pH-Wert in nur fünf von zehn Fällen erreicht wird, unabhängig davon, ob er sauer oder alkalisch ist, kann überprüft werden, ob die Menge an Säure bzw. Lauge angepasst werden muss, um gezielt in den gewünschten Bereich zu gelangen. Das wäre eine klassische Prozessoptimierung, die auf den ermittelten Daten aus der Qualitätskontrolle basiert.
Die Nutzung der Daten aus der Qualitätskontrolle ermöglicht somit eine gezielte Steuerung von Prozessen, um den angestrebten Output zu erreichen. Durch die regelmäßige Überprüfung und Anpassung der relevanten Parameter wird sichergestellt, dass das Endprodukt die gewünschten Qualitätsstandards erfüllt und den Anforderungen der Kunden entspricht.
# Reinigung von Mischbehältern
Eine weitere Anwendung der Inprozesskontrolle findet sich im Bereich der Reinigung. Nicht nur bei Produkten aus dem Lebensmittel- oder Pharmabereich müssen die Mischbehälter vorab gründlich gereinigt und die Reinigung dokumentiert werden. Dabei wird eine Probe entnommen und im Labor auf bestimmte Parameter kontrolliert. Erst nach erfolgreicher Kontrolle wird der Mischbehälter für die Produktion freigegeben.
Darüber hinaus werden von der Qualitätssicherung die Reinigungs-Checklisten, die von den Mitarbeitern im Zuge der Reinigung des jeweiligen Bereiches ausgefüllt werden, kontrolliert und archiviert.
3. Warenausgangskontrolle
Die Ausgangskontrolle spielt eine entscheidende Rolle, um sicherzustellen, dass die Ware vor dem Versand den erforderlichen Qualitätsstandards entspricht. Neben der optischen Kontrolle der Waren und der Sicherstellung der Chargenrückverfolgung gehören die Erstellung von Analysezertifikaten, die Versanddokumentation sowie im erweiterten Sinne auch der Ausgangscheck für Gefahrguttransporte zu diesem Aufgabenbereich:
# Chargenrückverfolgung
In unserem Mischbereich unterziehen wir jedes fertig gemischte Produkt einer umfassenden Bemusterung und Analyse auf Batch-Basis, bevor es abgefüllt wird. Von jedem Mischansatz wird ein Muster entnommen und gründlich untersucht.
Nach der Überprüfung wird die Mischung in Gebinde abgefüllt, wodurch mehrere Chargen entstehen. Jede dieser Chargen wird mit einer eindeutigen Chargennummer versehen, die ebenso im ERP-System abgebildet wird und sich auf die ursprüngliche Mischung bezieht.
Rückstellmuster dienen als Vergleichsmaterial für spätere Produktionen und können im Falle von Kundenreklamationen oder anderen Qualitätsproblemen zur Untersuchung herangezogen werden.
# Analysezertifikate 
Im Zuge der Ausgangskontrolle werden von der Qualitätskontrolle Analysezertifikate für unsere Kunden erstellt, die Informationen über die analysierten Parameter des Produkts liefern.
Die Zertifikate werden in Papierform ausgefertigt und den Lieferungen gemeinsam mit dem Lieferschein, der gleichzeitig das Gefahrgut-Beförderungspapier ist, beigefügt. Oft wird das Zertifikat auch automatisch über unsere Labordatenbank an den Kunden per E-Mail versendet. Diese Kunden erhalten das Analysezertifikat somit schon vor dem Eintreffen der Ware.
# Fotodokumentation
Als Teil der Ausgangskontrolle werden von jedem Gebinde, jeder Palette oder jedem IBC Fotos angefertigt, um den Zustand der Ware zu dokumentieren. Diese Fotos dienen als visueller Nachweis für die Qualität der Produkte vor der Auslieferung.
# Ausgangscheck für Gefahrguttransporte
Neben den zuvor genannten Aufgaben der Qualitätskontrolle sorgt die Qualitätssicherung im Rahmen der Ausgangskontrolle für die Durchführung der Checks von Gefahrguttransporten und die Überprüfung der LKWs:
- Prüfung der Verpackung und der Gefahrgutkennzeichnung (Etiketten, Sicherheitshinweise).
- Dokumentation und Überprüfung der Transportdokumente (Gefahrgut-Beförderungspapier, Analysezertifikate).
- Prüfung von Transportfahrzeugen und -ausrüstung (Inspektion der Fahrzeuge auf ihre Sicherheitsausstattung und die für Gefahrgut spezielle Notfallausrüstung).
Etwa 95 Prozent unserer LKWs transportieren Gefahrgut. Jede Mitarbeiterin und jeder Mitarbeiter der QK und QS durchläuft daher eine einwöchige, externe Schulung zum Gefahrgutbeauftragten, die alle 5 Jahre aufgefrischt wird.
Mustererstellung in der Qualitätskontrolle
Im Labor führen wir neben unseren regulären Aufgaben auch die Herstellung von Kundenmustern durch. Das bedeutet, dass wir entweder einzelne Rohstoffe bemustern, bei denen ein Mitarbeiter aus der Abteilung ein Muster von einem eingelagerten Behälter entnimmt, oder auch Mischungen für Kunden im Qualitätskontrolllabor herstellt.
Bei komplexeren Mischungen ist die Beteiligung der F&E-Abteilung erforderlich, da spezielle Anforderungen und Vorgaben berücksichtigt werden müssen. Bei einfacheren Mischungen oder Verdünnungen können die Muster direkt im Kontrolllabor hergestellt werden.
In einigen Fällen konzentriert sich die Mustererstellung lediglich darauf, bestimmte Parameter für Spezifikationen oder Sicherheitsdatenblätter zu ermitteln. Ein Beispiel dafür ist die Überprüfung des pH-Werts einer Mischung, um festzustellen, ob sie alkalisch, neutral oder sauer ist. Durch diese Analysen erhalten wir wichtige Informationen über die Eigenschaften der Mischung und können sicherstellen, dass sie den erforderlichen Standards entspricht.
Die Bestimmung solcher Parameter ist sowohl für die Qualitätssicherung als auch für die Erfüllung rechtlicher Anforderungen wichtig, insbesondere im Hinblick auf Sicherheitsaspekte.
Nahtlose Rückverfolgbarkeit: Die Rolle von QS und QK
Eine weitere wesentliche Aufgabe der QS und QK ist die lückenlose Verfolgung und Dokumentation aller relevanten Daten und Informationen entlang des gesamten Produktions- und Lieferprozesses. Die Aufgaben zwischen QK und QS sind wie folgt aufgeteilt:
- Aufgaben der Qualitätskontrolle: Sicherstellung der Nachvollziehbarkeit von Proben, Tests und Ergebnissen durch Aufzeichnungen über die Probenentnahme, die Analysemethoden, die verwendeten Geräte, die durchgeführten Tests und andere relevante Details.
- Aufgaben der Qualitätssicherung: Identifizierung und Kennzeichnung der Produkte (Chargen-/Seriennummer), Dokumentation von relevanten Daten und Informationen (Rohstoffe, verarbeitete Produkte, Verfahren, Produktionsprozess, Kundenlieferungen) sowie Festlegung von Aufzeichnungen und Rückverfolgbarkeitsketten.
Die Rückverfolgbarkeit ist für die Erfüllung gesetzlicher Anforderungen und Normen von hoher Relevanz, insbesondere im Hinblick auf die Produkthaftung. Sie ermöglicht es, im Falle von Reklamationen schnell zu reagieren und geeignete Maßnahmen zu ergreifen.
Effizientes Reklamationsmanagement bei der Donauchem
Das Reklamationsmanagement der Donauchem ist ein integraler Bestandteil des Qualitätsmanagementsystems (QMS) und wird von der QS und QK abgewickelt. Es umfasst verschiedene Aspekte wie Rohstoffe, Kundenreklamationen und interne Reklamationen:
1. Kundenreklamationen
Kundenreklamationen betreffen mitunter Transportschäden, wie zum Beispiel undichte Kanister, die während des Umschlag- und Transportvorgangs beschädigt wurden. Bei der Ursachenanalyse prüfen wir, ob die Schäden durch unsachgemäße Handhabung, schadhafte Paletten oder Stöße während des Transports entstanden sind. Durch den Vergleich der Fotos aus der Ausgangskontrolle mit den Bildern des Kunden können wir die Ursache des Schadens schnell identifizieren und angemessene Maßnahmen ergreifen.
Zusätzlich können auf der chemischen Seite Abweichungen vom gewünschten Qualitätsstandard auftreten, wie zum Beispiel Verunreinigungen. In solchen Fällen spielt die Qualitätskontrolle eine entscheidende Rolle, indem sie durch den Rückgriff auf Laborwerte oder den Vergleich des reklamierten Produkts mit dem Rückstellmuster feststellt, ob das Produkt tatsächlich von der erwarteten Qualität abweicht.
Es kommt immer wieder auch zu Reklamationen aufgrund spezifischer Eigenschaften des Produkts, die jedoch manchmal gar keinen eindeutigen Mangel darstellen. Ein gutes Beispiel dafür sind Verfärbungen: Das Produktdatenblatt beschreibt das Produkt beispielsweise als „wasserklare bis gelbliche Lösung“. Wenn der Kunde nun mehrmals ein wasserklar erscheinendes Produkt erhält, aber einmal eine gelbliche Lösung, könnte dies zu einer Reklamation führen, obwohl das Produkt innerhalb der festgelegten Spezifikationen liegt.
Bei der Donauchem haben wir für jede Art von Kundenreklamation die Verantwortlichkeiten benannt, damit innerhalb eines festgelegten Zeitrahmens eine Stellungnahme an den betroffenen Kunden erfasst wird. Diese beinhaltet den Verlauf des Problems und die ergriffenen Maßnahmen zur Verbesserung. Durch diesen Ansatz optimieren wir kontinuierlich unsere Prozesse und erfüllen die Anforderungen der ISO 9001.
|
Produktdatenblatt
|
Sicherheitsdatenblatt
|
Das Produktdatenblatt (PDB) dient in erster Linie dazu, dem Kunden Informationen über das Produkt und seine Eigenschaften zur Verfügung zu stellen.
Das PDB enthält technische Daten wie physikalische und chemische Eigenschaften, Anwendungshinweise, Lagerungsempfehlungen und Informationen zur Handhabung.
|
Das Sicherheitsdatenblatt (SDB) konzentriert sich auf die Sicherheit und den Umgang mit dem Produkt. Es dient als grober Richtwert und ist in der Regel auf Angaben zu den verwendeten Rohstoffen beschränkt.
Das SDB liefert detaillierte Informationen über die potenziellen Gefahren, die von einer Substanz oder einem Gemisch ausgehen können, sowie über Maßnahmen zu Vermeidung von Risiken und zur Ersten Hilfe im Notfall. |
Abbildung 5: Der Unterschied zwischen Produkt- und Sicherheitsdatenblatt.
2. Interne Reklamationen
Auch interne Reklamationen werden im Rahmen des Reklamationsmanagements berücksichtigt. Wenn es zu Abweichungen innerhalb des Unternehmens kommt, die den ordnungsgemäßen Ablauf von Prozessen, Arbeitsabläufen oder Ergebnissen beeinflussen, werden diese als interne Reklamationen behandelt.
3. Rohstoffreklamationen
Im Hinblick auf Rohstoffe können Reklamationen auftreten, wenn gelieferte Waren nicht den erforderlichen Spezifikationen entsprechen oder Transportschäden aufweisen. Die Überprüfung der Rohstoffe erfolgt wie bereits beschrieben von der QK im Zuge der Eingangskontrolle.
Bei der Bearbeitung von Rohstoffreklamationen ist nach Absprache mit Einkauf, Transporteur und Lieferant die weitere Vorgangsweise abzustimmen.
Fazit: Qualität entlang der Wertschöpfungskette
Qualitätskontrolle und Qualitätssicherung sind für die chemische Distribution und Lohnproduktion von zentraler Bedeutung und untrennbar miteinander verknüpft. Unsere Qualitätskontrolle überwacht und prüft die Produktqualität, während die Qualitätssicherung die notwendigen Prozesse und Verfahren definiert, um diese Qualität entlang der gesamten Wertschöpfungskette zu gewährleisten. Als wesentliche Elemente unseres Qualitätsmanagementsystems tragen sie maßgeblich dazu bei, dass wir Produkte von hoher Qualität und Sicherheit unseren Kunden anbieten und liefern können.
Wir freuen uns darauf, Sie auch in Zukunft mit unseren Produkten zu unterstützen. Bei Anfragen sind wir jederzeit für Sie da -
kontaktieren Sie uns noch heute.
 www.donauchem.at
www.donauchem.at