Nicht nur ist Wasser unser wichtigstes Lebensmittel, sondern viele Lebensformen auf unserer Erde bestehen zu einem großen Teil aus Wasser. Der menschliche Körper etwa enthält zwischen 50-65% Wasser, viele höhere Tiere haben einen ähnlich hohen Wasseranteil in ihrem Körper.
Die wichtigste aller Flüssigkeiten erfüllt gleich mehrere lebenswichtige Funktionen, die die Basis für den Kreislauf des Lebens bilden. Denn durch die Eigenschaften des Wassers können in der Natur viele Vorgänge überhaupt erst stattfinden.
Wasser als chemische Verbindung
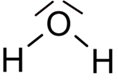
Wasser besitzt die chemische Formel H
2O, bestehend aus den Elementen Sauerstoff (O) und Wasserstoff (H).
Als Wasser wird es in flüssigem Aggregatzustand bezeichnet, als Wasserdampf im gasförmigen und Eis bezeichnet den festen Zustand von Wasser. Natürlich vorkommendes Wasser ist kein reines Wasser, denn Anteile von Mineralstoffen, Salzen, Gasen und organischen Verbindungen finden sich gelöst darin.
Alle biologischen Vorgänge auf unserem Planeten laufen nur Dank flüssigem Wasser ab.
Strukturformel:
Grundbaustein des Lebens
Das Leben ist nach dem heutigen Erkenntnisstand im Wasser entstanden. Autotrophe Schwefelbakterien (Prokaryoten) produzieren aus Schwefelwasserstoff und Kohlendioxid unter Zufuhr von Lichtenergie organische Kohlenstoffverbindungen und Wasser:
18 H
2S + 6 CO
2 
C
6H
12 + 12 H
2O + 18 S
Als Nachfolger produzierten Blaubakterien (Cyanobakterien) und autotrophen Eukaryoten unter Zufuhr von Licht aus Wasser und Kohlendioxid Traubenzucker und Sauerstoff:
6 CO
2 + 12 H
2O

C
6H
12O
6 + 6 O
2 + 6 H
2O
Durch diesen Prozess reicherte sich im Wasser und in der Atmosphäre immer mehr Sauerstoff an. Damit wurde die Gewinnung von Energie durch Zellatmung (Dissimilation) möglich:
C
6H
12O
6 + 6 O
2 
6 H
2O + 6 CO
2
Voraussetzung für die Fähigkeit, mit dem giftigen Sauerstoff (Oxidation der empfindlichen Biomoleküle) umzugehen, waren Enzyme wie die Katalase, die eine strukturelle Ähnlichkeit mit dem Sauerstoff transportierenden Hämoglobin aufweist. Aerobe Purpurbakterien nutzten vielleicht als erstes den giftigen Sauerstoff zum energieliefernden Abbau von organischen Stoffen. Nach der Endosymbiontentheorie nahmen damals noch anaerobe Eukaryoten die aeroben Prokaryoten (wahrscheinlich Purpurbakterien) auf.
Wasser wurde damit zum Medium grundlegender biochemischer Vorgänge (Stoffwechsel) zur Energiegewinnung und -speicherung:
- Photosynthese, Dissimilation
- Glykolyse
- Zitronensäurezyklus
- Fettabbau
- Eiweißabbau
- Harnstoffzyklus
Eigenschaften von Wasser
Wasser kommt in zwei Isomeren (para- und ortho-Wasser) vor, die sich im Kernspin der beiden Wasserstoffatome unterscheiden.
Wasser bildet ein sogenanntes Dipol, da Sauerstoff eine höhere Elektronennegativität besitzt als Wasserstoff. Durch die Dipol-Bildung besitzen Wasserstoffmoleküle ausgeprägte zwischenmolekulare Anziehungskräfte und lagern sich über Wasserstoffbrückenbildungen zu Clustern zusammen.
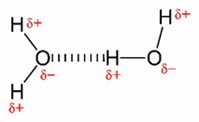
Abb.: Darstellung zweier Wassermoleküle mit rot markierten Partialladungen, verbunden durch eine gestrichelt gezeichnete Wasserstoffbrückenbindung. Quelle:
Wikipedia
Wissenswertes zu Wasser
- Es hat eine Dichte von rund 1000 kg/m³ (ursprünglich die Definition des Kilogramms), genauer: 999,975 kg/m³ bei 3,98 °C. Als Dichteanomalie bezeichnet man die auf der Wasserstoffbrückenbindung beruhende Eigenschaft, dass Wasser bei dieser Temperatur die höchste Dichte hat und beim Abkühlen unter diese Temperatur kontinuierlich und beim Gefrieren sogar sprunghaft an Volumen zunimmt, also an Dichte verliert, so dass Eis auf Wasser schwimmt,
- Es hat eine Viskosität von 1,0019 mPa·s (0,010019 Poise) bei 20 °C,
- Es hat eine der höchsten spezifischen Wärmekapazitäten von Flüssigkeiten bei Raumtemperatur (75,366 J·mol−1·K−1 entsprechend 4,18 kJ·kg−1·K−1 bei 20 °C,
- Es hat eine der größten Oberflächenspannungen aller Flüssigkeiten (nur Quecksilber hat eine noch größere); bei Wasser beträgt sie in feuchter Luft 72 mN/m bei +20 °C, so dass die Tröpfchenbildung erleichtert wird,
- Es hat eine der größten spezifischen Verdampfungsenthalpien aller Flüssigkeiten (44,2 kJ/mol entsprechend 2453 kJ/kg bei 20 °C; daher rührt der kühlende Effekt bei der Transpiration) sowie eine hohe Schmelzenthalpie (6,01 kJ/mol entsprechend 333 kJ/kg; so dass Salzwasser eine nur geringe Gefrierpunktserniedrigung im Vergleich zu reinem Wasser zeigt),
- Es hat eine geringe Wärmeleitfähigkeit (0,6 W/(m K) bei 20 °C).
- Je nach Isotopenzusammensetzung des Wassermoleküls unterscheidet man normales „leichtes Wasser“ (zwei Atome Wasserstoff: H2O), „Halbschweres Wasser“ (ein Atom Wasserstoff und ein Atom Deuterium: HDO), „schweres Wasser“ (zwei Atome Deuterium: D2O) und „überschweres Wasser“ (zwei Atome Tritium: T2O), wobei mit HTO und DTO noch weitere Moleküle mit gemischten Isotopen vorkommen.
- Wasser kann unter Hochspannung eine Wasserbrücke zwischen zwei Glasgefäßen ausbilden.
Wasserchemie
Hier werden die Eigenschaften des Wassers, seine Inhaltstoffe, die jeweiligen Umwandlungen des Wassers sowie der Stoffhaushalt von Gewässern beschrieben. Außerdem werden auch Reaktionen von Wasser bzw. durch Wasser hervorgerufene Reaktionen und der Zusammenhang mit diversen Wasserkreisläufen beschrieben.
Wasser ist ein Lösemittel für viele hydrophile gasförmige Substanzen, hydrophile organische Stoffe, Mineralien, diverse Ionenverbindungen und andere Stoffe.
Generell unterscheidet man folgende Wassertypen in der Wasseranalytik:
- Trinkwasser
- Mineralwasser
- Heilwasser
- Tafelwasser
- Süßwasser/Meerwasser/Salzwasser/Brackwasser
- Reinstwasser
- Demineralisiertes Wasser
- Destilliertes Wasser
- Enteisentes Wasser
- Prozesswasser
- Nutzwasser
- Abwasser, (Haushalts-Abwässer, landwirtschaftliche Abwässer, Industrie-Abwässer)
- Regenwasser
- Grundwasser
- Oberflächenwasser (Fließ- und Stehgewässer)
Wasser als wichtigste Ressource
Gäbe es in der Atmosphäre unseres Planeten kein Wasser, wäre die Durchschnittstemperatur auf der Erde viel niedriger, denn die Wärme der Sonne würde direkt ins Weltall reflektiert werden.
Der Wasseraustausch in Gewässern entsteht durch Temperaturschwankungen, die der jeweiligen Jahreszeit geschuldet sind. Und auch die Verteilung von Wärme auf der Erde fußt auf dem Faktor, dass große Meeresströmungen warmes Wasser in kältere Gebiete bringen. Alles Wasser auf der Erde ist in einem stetigen Kreislauf und kann daher weder verbraucht noch neu erschaffen werden.
Um die grundlegende Bedeutung von Wasser hervorzuheben, wurde es 2005 in einer Resolution des EU-Parlaments als fundamentales Menschenrecht definiert. Leider steht es jedoch nicht allen Menschen in ausreichender Menge und Qualität zur Verfügung.
Vor allem Industrieländer sind dabei, eine verbesserte Nutzung ihrer Wasserressourcen umzusetzen und dadurch den Wasserverbrauch minimieren. Die Kehrseite dieser Entwicklung ist jedoch, dass Produktionsstätten in andere Länder verlagert werden, wodurch sich sowohl der Wasserverbrauch für diese Produktionen als auch die damit einhergehenden Umweltbelastungen in diese Länder verlagern.
Wasserkreislauf
Der Wasserkreislauf beginnt, wenn Wasser aus den Meeren verdunstet. Der Wasserdampf geht anschließend je nach Region und Jahreszeit in Form von Regen, Hagel oder Schnee wieder auf die Erdoberfläche nieder. Letzten Endes fließt es in den Flüssen wieder ins Meer zurück - der Kreislauf beginnt von vorne.
Jede Sekunde verdunstet auf unserem Planeten etwa eine Milliarde Liter Wasser. Trotzdem nehmen nur 0,77 Prozent des Gesamtwassers am Wasserkreislauf teil. Der Großteil ist in Ozeanen oder Eiskappen, gebunden.
Was passiert mit Niederschlagwassers?
- Im oberirdischen Wasserkreislauf fließt Niederschlagswasser meist als Oberflächenwasser in Flüssen in das Meer, wo es an der Oberfläche wieder verdunstet.
- Der unterirdische Wasserkreislauf wird von dem Wasser gespeist, das tiefer in den Boden eintritt. Hierzu zählt auch das Grundwasser.
- Wasser aus Niederschlag wird außerdem von Pflanzen aufgenommen und mittels Transpiration wieder in die Atmosphäre abgegeben.
- Auch verdunstet ein Teil des Wassers an der Oberfläche, bevor es in einen anderen Wasserkreislauf eintreten kann.

Abb.: Der Wasserkreislauf - Quelle:
Wasserwerk.at
Wassernutzung
Wasser begleitet uns fast allen Bereichen des Lebens.
Ob als notwendiges Lebensmittel oder für das Wachstum von Pflanzen und somit Nahrung erforderlich, hat es auch in der Industrie eine nicht zu unterschätzende Rolle.
Als Reinigungsmittel, Kühlmittel, Produktionsmittel, als Energielieferant oder als Transportmittel kommt es in diversen Anwendungsbereichen zum Einsatz. Doch gerade hier gilt es, besonders darauf zu achten, dass das Wasser wieder in sauberem Zustand in den natürlichen Kreislauf zurückgeführt wird. Verschmutzungen müssen mittels unterschiedlicher Filtriermethoden vor dem Einleiten entfernt werden, um Umweltfolgen wie Fischsterben zu vermeiden. Und auch die Temperatur dieses Wassers darf den natürlichen Kreislauf nicht stören.
Grundwasserentnahme nach Sektoren
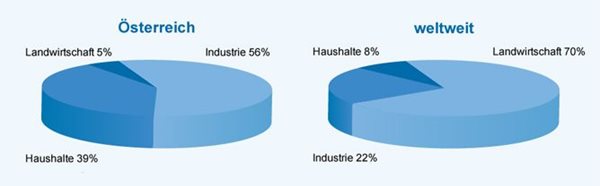
Quelle:
Wasserwerk.at
Wasser als Lösungsmittel
Wasser ist eines der wichtigsten Lösungsmittel. Denn aufgrund seiner Polarität und der Fähigkeit, Wasserstoffbrückenbindungen zu bilden, kann Wasser viele Arten von Molekülen lösen. Diese Fähigkeit ist die Basis für diverse chemische Reaktionen.
Auch im Alltag verwenden wir Wasser als Lösungsmittel: etwa bei der Zubereitung von Tee oder Kaffee, die heißem Wasser Aroma und Farbe geben. Oder dem Beigeben von Zucker in diese Tasse Tee, der sich vollständig im Wasser auflöst.
Die meisten dieser Reaktionen, die wichtig für das Leben sind, laufen in einer wässrigen Umgebung im Inneren der Zellen ab. Im Körper übernimmt Wasser die Funktion eines Transportmittels, denn es löst Nährstoffe und andere lebenswichtige Stoffe und transportiert diese durch unseren Körper. Für Wasserlebewesen etwa ist es lebensnotwendig, dass sich Sauerstoff in Wasser löst, den sie über ihre Kiemen oder direkt über die Haut aufnehmen.
Quelle:
Klassewasser.de
Synthese, Elektrolyse und chemische Verwendung
Als chemische Verbindung wurde Wasser erstmals im 18. Jahrhundert synthetisiert. Es wurde ein Gemisch aus Wasserstoff und Luft dabei zur Explosion gebracht (Knallgas-Reaktion).
Durch die Elektrolyse von Wasser erhält man Wasserstoff, der als wichtiger zukünftiger Energieträgen gilt. Dabei wird Wasser im Hofmannschen Wasserzersetzungsapparat in seine Bestandteile zerlegt. Reaktionsschema:
2 H
2O

2 H
2 + O
2
Warum entstehen Bläschen in siedendem Wasser?
Durch Wärmeeinwirkung bewegen sich die Wassermoleküle schneller. Werden 100 °C erreicht, wird der Aggregatszustand von flüssig aufdampfförmig geändert. Dampf hat ein um etwa 1600-fach höheres Volumen sowie eine geringere Dichte: Das Wasser beginnt zu sieden, wobei die Dampfblasen von Schichten noch nicht so heißen Wassers abgekühlt werden und wieder zu flüssigem Wasser kondensieren. Erreicht schließlich die gesamte Wassermenge die Temperatur von 100 °C, so gelangen die nun großen Dampfblasen bis an die Oberfläche: Das Wasser kocht.
Druck und Temperatur sind die bestimmenden Faktoren für die Löslichkeit von Gasen im Wasser. Gasbläschen, die bereits bei geringfügiger Erwärmung sichtbar werden, bestehen aus gelösten Gasen. Ursache ist die geringere Wasserlöslichkeit von Gasen bei Erwärmung. Wasser, das sich eine Zeit lang in einer unter Druck stehenden Leitung oder Flasche befunden hat, hat oft einen Überschuss an Gasen gelöst. Daher reicht schon das Wegnehmen des äußeren Drucks, dass sich – bevorzugt an Keimen an der Wandung – Gasblasen ausscheiden und bis zu einer Größe von 1–2 mm auch haften bleiben.
Wasser in der Technik und darüber hinaus
Zur Verwendung in der Technik hat Wasser, meist in flüssigem Zustand, manchmal auch in Form von Wasserdampf oder Eis, eine sehr hohe Bedeutung.
- Die Gewichtskraft von Wasser wird in Wassermühlen und Wasserturbinen zur Gewinnung von mechanischer oder elektrischer Energie genutzt.
- Dampf wird in Dampfturbinen, die in vielen modernen Kraftwerken vorkommen, genutzt, um die primär erzeugte Wärme zunächst in mechanische Energie und schließlich mit einem Generator in elektrische Energie umzuwandeln. Die (Kolben-)Dampfmaschine war vor allem in der industriellen Revolution von Bedeutung.
- Eismaschinen dienen der Herstellung und Verarbeitung von Speiseeis und Eiswürfeln. Die Eisbearbeitungsmaschine ermöglicht das Glätten von Kunsteisbahnen.
- Bei der Wärmeübertragung dient Wasser als Transportmittel für Heizungen oder in der Wasserkühlung. Auch die Erzeugung von Kälte durch Verdunstung ist durch Wasser möglich. Kältemaschinen funktionieren auf Basis der Adsorption von Ammoniak in Wasser oder Wasserdampf in (wässriger) Lithiumbromid-Lösung.
- Wasser dient kalt und warm zum waschen und reinigen, oft in Verbindung mit Detergenzien, Laugen oder Säuren. Außerdem ermöglicht es das Lösen (Auslaugen von Salzlagerstätten), Trennen über Chromatographie oder Extrahieren (Aufgüsse), das Umkristallisieren (Abbinden von Gips oder Zement zusammen mit Kohlenstoffdioxid. Auch in Form eines Druckstrahls kann es zum Hochdruckreinigen oder aber zum Wasserstrahlschneiden, etwa in hygienesensiblen Bereichen, verwendet werden.
- In Form von Gel wird Wasser als Schallübertragungsmedium vom Sensorkopf zum menschlichen Körper bei der Ultraschalldiagnostik genutzt. Wasser ist Schallübertragungsmedium beim Echolot.
- Als Medium mit hoher Oberflächenspannung und guter Verdunstungsrate dient Wasser zum verschieblichen Anklatschen von Beschriftungsfolie auf Schaufenstern, Autokarosserien und anderen glatten zu kaschierenden Oberflächen, ebenso als Gleit- und Dichtmittel für Saugnäpfe. Die Oberflächenspannung von Wasser erlaubt im Zusammenspiel mit Seife Seifenblasen und das Bauen von Schichten ab Moleküldicke und feiner Membrane für physikalische Experimente. Der Wasserläufer kann mittels Dellen in der Oberfläche laufen, Biofilme können sich ausbreiten.
- Ursprüngliche Hydraulik verwendet Wasser als Druckübertragungsmedium sowie als Fontänen in Springbrunnen und Wasserspielen, die auch Verdunstungskühlung und Lichteffekte ermöglichen. Das Aufbrechen von geologischen Schichten beim Fracking ist ebenfalls eine Hochdruckanwendung.
- Durch Wasser erzeugter Auftrieb erlaubt Lebewesen, Schiffen und Bojen das Schwimmen. Ballasttanks helfen, unbeladene oder ungleichmäßig beladene Schiffe zu stabilisieren. Bei U-Booten ermöglichen sie ein Auf- und Abtauchen. Auch im Bereich der Seilbahnen gibt es Anwendungen, wenn diese Lifte im Gegenzug von Wasserballasttanks gezogen bzw. gehoben werden.
- Wasser als Dissoziationsmedium dient der Elektrolyse, Galvanik, Akku- und Batterietechnik sowie in alten Kraftwerken als Strom-Regelungstank. Weiters kommt Wasser als Lösungsmittel in der wässrigen Chemie zum Einsatz.
- In der Medizin dient Wasser als lösendes Medium zum Injizieren oder Infundieren von Stoffen in den Körper, um den Wasserhaushalt des Körpers zu korrigieren, zum Aufweichen sowie zum Spülen.
- Wasser ermöglicht durch Einlegen das Biegsammachen von Weidenruten, Peddigrohr etc. Hartholz wird unter Wasserdampf zu Bugholzmöbeln geformt.
- Wasser kann Infrarotstrahlung aus Glühlampenlicht ausfiltern und absorbiert ionisierende Strahlung im Abklingbecken von Kernkraftwerken.
- In Wasserwerfern wird Wasser, mit und ohne chemischen Zusatz, als Munition eingesetzt.
- Reinstwasser leitet Strom schlecht. Erst wenn andere Stoffe, die sich in Ionen auflösen können, hinzukommen, kann es elektrischen Strom übertragen.
- In Kernkraftwerken wird Wasser als Moderator genutzt, d. h., um Neutronen abzubremsen.
Wasseraufbereitung
Stickstoffverbindungen, Schwermetalle, Salze und Kohlenwasserstoffe, die bereits im Oberflächenwasser enthalten waren, sind oft in gefördertem Rohwasser nachzuweisen. Da diese Stoffe gesundheitsschädlich sind, müssen sie durch diverse Verfahren entfernt werden.
Danach muss das Wasser durch spezielle Filter geleitet werden, die mit Aktivkohle gefüllt sind. Aktivkohle kann an sehr großen Oberflächen Stoffe anlagern und so aus dem Wasser entfernen.
Erst jetzt darf das Wasser als Trinkwasser bezeichnet werden. Vor dem Einspeisen in das Versorgungsnetz wird es falls erforderlich sicherheitshalber desinfiziert.
Die Sparte Wassertechnik der Donau Chemie AG entwickelt, produziert und verkauft Fällungs- und Flockungsmittel auf Basis von Eisen- und Aluminiumchloriden für die breit gefächerte Behandlung von Wasser sowie Produkte für spezielle industrielle Anwendungen. Das Angebotspektrum reicht hier von der kommunalen bzw. industriellen Abwasserbehandlung über die kommunale Trink- und Badewasseraufbereitung bis hin zu Lösungen für die Papierindustrie sowie zu Produkten zur Optimierung von Biogasprozessen.
Wasser und der Klimawandel
Laut einer Studie der EU-Kommission haben Dürren in der Europäischen Union in den vergangenen 30 Jahren zugenommen. Waren davon bislang Hauptsächlich Süd- und Südosteuropa betroffen, werden zukünftig auch Gebiete in Mittel- und Osteuropa stärker unter Wasserknappheit leiden. Hier gilt es, die Wasserversorgung zu sichern und der Verschwendung von Wasser gegenzusteuern. Die Basis bildet die EU Wasserrahmenrichtlinie aus dem Jahr 2000. Sie schreibt vor, dass die Mitgliedstaaten bis 2010 Wasser effizienter nutzen müssen.
Klimaprognose für Österreich
Auch in Österreich muss Wasser jedem in guter Qualität zur Verfügung stehen, eine Verschwendung ist grundsätzlich zu vermeiden. Bereits jetzt machen sich
die Auswirkungen von ausbleibenden Regenfällen und
Dürren deutlich bemerkbar.
Laut Prognosen der Klimaforscher wird die Jahressumme der Niederschläge in Österreich zwar gleich bleiben, der Süden aber weniger davon abbekommen als das Gebiet nördlich des Alpenhauptkamms. Im Winter werden mehr Niederschläge in Form von Regen und weniger in Form von Schnee erfolgen, sodass weniger Grundwasservorräte gebildet werden können. Die Gletscher werden weiter zurückgehen. Der Süden, speziell Kärnten und die Oststeiermark, werden fast durchgehend von einer Abnahme der Niederschläge betroffen sein.
Fazit: Wasser ist auch in Österreich kein unerschöpfliches Gut
Wasser ist als Grundlage allen Lebens ein kostbares Gut, mit dem wir sorgsam umgehen müssen. Durch sparsamen Umgang im privaten wie industriellen Bereich sowie durch professionelle Wiederaufbereitung können Privatpersonen und Unternehmen dazu beitragen, die Wasserqualität auch für kommende Generationen sicherzustellen.
Eine globale Lösung ist aufgrund der steigenden Bevölkerungsdichte sowie der Industrie-, Gewerbe- und Landwirtschaftsentwicklung der letzten Jahrzehnte notwendiger denn je.
Für genauere Informationen zu unseren Produkten für die Behandlung und Aufbereitung
von Trinkwasser, Badewasser und Abwasser sowie zur Geruchs- & Korrosionsbekämpfung können Sie uns
jederzeit kontaktieren.
 www.donauchem.at
Quellenangaben:
www.donauchem.at
Quellenangaben:
https://www.derstandard.at/story/2000137296077/seen-bei-wiener-neustadt-statt-wasser-haben-wir-stinkenden-heilschlamm (Stand: 9.3.2023)
https://kurier.at/wissen/wissenschaft/duerre-die-wasserversorgung-wird-selbst-in-oesterreich-zum-problem/402304493 (Stand 9.3.2023)
http://www.wasserwerk.at/home/alles-ueber-wasser (Stand 9.3.2023)
https://klassewasser.de/content/language1/html/3697.php (Stand 9.3.2023)
http://wasseraktien.de/wasser/wasser-als-lebensgrundlage (Stand 9.3.2023)
https://www.umweltbundesamt.de/daten/wasser (Stand 9.3.2023)